
Buprenorphine was approved by the FDA for opioid use disorder nearly 25 years ago. Much of the information online is based on outdated information.
Both policies and dosing of buprenorphine continue to change to curb rising opioid overdose deaths caused by a surge of stronger opioids like fentanyl and carfentanil.
Table of Contents
Today, when someone says “Subutex,” they are usually referring to generic buprenorphine-only tablets. The Subutex brand has not been sold in the U.S. for more than a decade.
Indivior is the manufacturer behind both Subutex and Suboxone (buprenorphine + naloxone). For most patients, the naloxone version is ideal to reduce misuse. Buprenorphine-only is only used in specific cases like an allergy to naloxone.
Medication
Generic
Use
Buprenorphine
Subutex
Belbuca
Generic (generics produced by two manufactures)
Brand name
Brand name
Used only situationally (i.e. allergy/intolerance, pregnancy, inpatient setting, long-acting opioid withdrawals like methadone, etc)
Discontinued in 2011
Used only for chronic pain and is not FDA-approved for OUD treatment
Medication
Buprenorphine
Generic
Generic (generics produced by two manufactures)
Use
Used only situationally (i.e. allergy/intolerance, pregnancy, inpatient setting, long-acting opioid withdrawals like methadone, etc)
Medication
Subutex
Generic
Brand name
Use
Discontinued in 2011
Medication
Belbuca
Generic
Brand name
Use
Used only for chronic pain and is not FDA-approved for OUD treatment
2 mg

2 mg
Both buprenorphine and buprenorphine with Naloxone were approved for opioid use disorder in 2002.
Naloxone was added to discourage misuse. The extra medication does basically nothing when taken correctly, but triggers withdrawals instead of the “high” if the medication is injected.
Over time, that diversion-safety advantage became the default choice for most outpatient treatment and is often used even in inpatient settings too.
Zubsolv was FDA-approved in 2013 to give alternative dosing options that can often be lower because it is better absorbed.
Medication
Generic
Use
Brand name (generics also widely available)
Brand name
Brand name
Brand name
Used only situationally (i.e. allergy/intolerance, pregnancy, inpatient setting, long-acting opioid withdrawals like methadone, etc)
Different dosing options to give more precise or lower dosing compared to Suboxone (1.4mg and 5.7mg vs the standard 2mg and 8mg) and higher bioavailability (so less medication is needed)
Discontinued in 2020
Discontinued in 2019
Medication
Suboxone
Generic
Brand name (generics also widely available)
Use
Used as a take-home prescription or for inpatient detox
Medication
Zubsolv
Generic
Brand name
Use
Different dosing options to give more precise or lower dosing compared to Suboxone (1.4mg and 5.7mg vs the standard 2mg and 8mg) and higher bioavailability (so less medication is needed)
Medication
Bunavail
Generic
Brand name
Use
Discontinued in 2020
Medication
Cassipa
Generic
Brand name
Use
Discontinued in 2019
2mg/0.5mg

8mg/2mg
2mg/0.5mg
8mg/2mg

1.4mg/0.36mg

5.7mg/1.4mg

2mg/0.5mg

8mg/2mg
Long-acting buprenorphine is a newer way to deliver the same medication without needing a daily tablet or film.
The 6-month implant approved in 2016 has since been discontinued. But, Sublocade from 2017 and Brixadi from 2023 support treatment adherence, especially when fentanyl is involved. ⓘ
Long-acting buprenorphine is much less common than tablets/films and insurance coverage is often more challenging.
Medication
Generic
Use
Sublocade
Brixadi
Probuphine
Brand name for a monthly injection
Brand name for a weekly or monthly injection
Brand name for a 6 month implant
Prevents missed dosing by mistake or on purpose
Requires less commitment, encourages check-ins with the medical provider
Discontinued in 2020
Medication
Sublocade
Generic
Brand name for a monthly injection
Use
Prevents missed dosing by mistake or on purpose
Medication
Brixadi
Generic
Brand name for a weekly or monthly injection
Use
Requires less commitment, encourages check-ins with the medical provider
Medication
Probuphine
Generic
Brand name for a 6 month implant
Use
Discontinued in 2020
In February 2025, Sublocade’s label was updated to support faster starts for the injections. A patient does not have to be on buprenorphine for a week before the first Sublocade shot. Plus, the shot can now be injected in more areas.
These are separate medications that do not contain buprenorphine, but are often compared and confused.
Medication
Pros Compared to Buprenorphine
Cons Compared to Buprenorphine
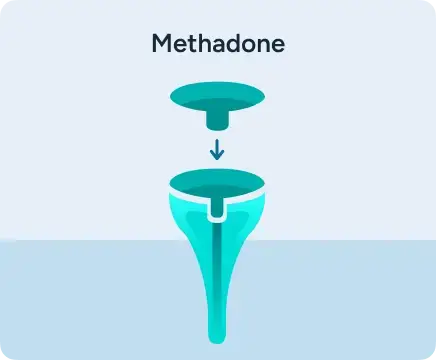
Methadone
Naltrexone
Strong
Non-opioid No dependence (no taper required to stop) Monthly injection also available (Vivitrol)
Carries a higher risk for abuse and overdose
Only available through Opioid Treatment Programs (OTPs / methadone clinics)
Longer commitment (12 months+ recommended)
Does not help with withdrawals / detox
Medication
Methadone
Pros Compared to Bupenorphine
Strong
Cons Compared to Bupenorphine
Carries higher risk for abuse and overdose Only available through Opioid Treatment Programs (OTPs / methadone clinics) Longer commitment (12 months+ recommended)
Medication
Brixadi
Pros Compared to Bupenorphine
Non-opioid No dependence (no taper required to stop) Monthly injection also available (Vivitrol)
Cons Compared to Bupenorphine
Does not help with withdrawals / detox
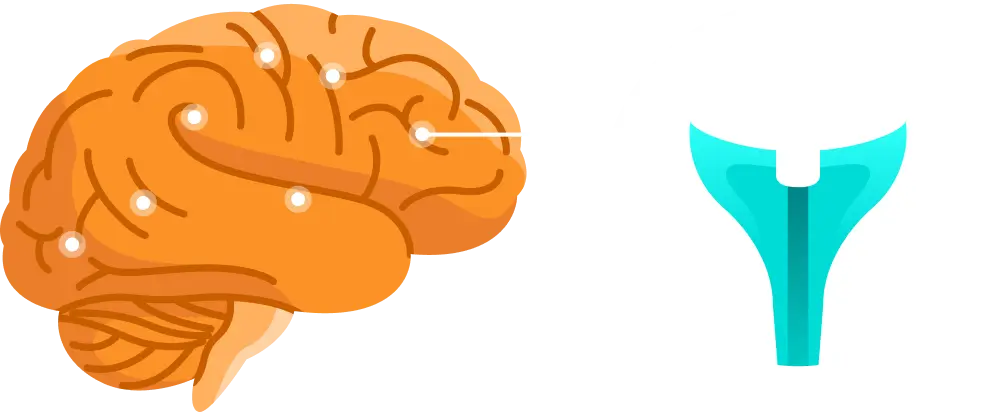
Full agonist:
generates effect
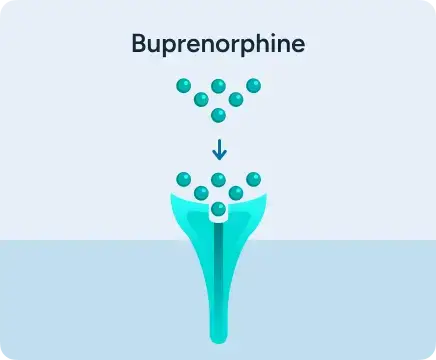
Partial agonist:
generates limited effect
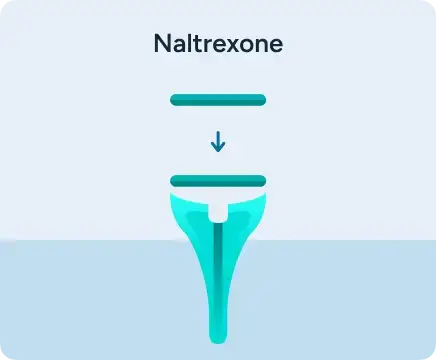
Antagonist:
blocks effect
Previously, even though buprenorphine is FDA-approved for opioid use disorder (OUD), clinicians had to get a special federal permission called the DATA 2000 “X-waiver,” and were limited by patient caps (often starting at only 30 patients). The Consolidated Appropriations Act, 2023 (MAT Act provision) removed those federal restrictions.
In 2026, most physicians (MD/DO) can prescribe take-home buprenorphine (including hospitals, primary care, psychiatrists, and addiction treatment programs). Nurse practitioners and physician assistants can also prescribe in many states.
When more clinicians can prescribe, buprenorphine becomes easier to start and easier to stay on. It also reinforces a simple truth: opioid use disorder is a medical condition, and addiction treatment belongs in regular healthcare.
Medical doctors (MD/DO), physician assistants (PA) and nurse practitioners (NP) already maintain a DEA registration to prescribe medications.
Beginning June 2023, clinicians renewing or applying for a DEA registration have to attest to a one-time training (usually 8 hours) on the treatment and management of substance use disorders (MATE Act requirement).
This is how access expanded without the x-waiver while keeping safety training in place.
A study that tracked data through the end of 2023 found that more clinicians began prescribing buprenorphine after the X-waiver was removed, but the total number of patients receiving buprenorphine did not increase much. ⓘ
This suggests the biggest barriers are now practical, like clinic capacity, follow-up availability, stigma, and pharmacy or insurance issues.
As of January 1, 2026, a DEA/HHS rule allows buprenorphine for opioid use disorder to be started through telemedicine, including video and phone-only visits.
Patients are now able to use telehealth to:
Most states (including FL, MA, OK, NJ, and AZ) can accommodate the new federal rules. In certain states (like AR), state laws need to be updated to to start buprenorphine without an in-person visit even though it’s federally allowed. In other states (like IN) video is required, so the phone-only portion is not initially allowed.
For many patients, telehealth can start sooner and helps transportation, work schedules, childcare, or distance makes frequent office visits hard. Telehealth access can also help patients begin MAT detox sooner when medication-assisted treatment is recommended as part of a supervised recovery plan.
Prescription Drug Monitoring Programs (PDMPs) are state-run databases that track controlled-substance prescriptions (like opioid pain meds, stimulants, benzodiazepines, and buprenorphine). Clinicians and pharmacists can see what controlled meds have been filled recently to prevent duplicate prescriptions or dangerous interactions.
Under the federal tele-buprenorphine rule, the prescriber generally has to check the PDMP for the state where the patient is located before prescribing, and pharmacies have an added identity-verification step before filling. PDMP is not new, but is becoming more relied on with other safeguard restrictions being lifted.
The biggest change in 2026 is clearer guidance that buprenorphine dosing should be individualized, not forced into one standard.
Fentanyl is extremely potent, and many patients arrive with a higher tolerance, faster withdrawal swings, and more fear about starting buprenorphine.
More complexity starting buprenorphine
Fentanyl is extremely potent and can linger in the body, so the “right time” for the first dose of buprenorphine is not always predictable. Clinicians spend more time assessing withdrawal symptoms and adjusting dosing person-by-person.
Microinductions (The Bernese Method)
Microinductions aren’t new, but became a hot topic in the fentanyl era.
Buprenorphine is started in tiny doses and slowly increased with close monitoring to lower the risk of precipitated withdrawal.
Microinductions work well at our The Haven Detox inpatient settings where medical staff is watching and adjusting 24/7.
However, despite the online hype, outpatient results for microdosing buprenorphine are lax. A 2025 JAMA study of patients using fentanyl found low success and retention rates with microdosing.ⓘ Patients often struggle without the medical oversight to balance dosing with real-time symptoms and other detox medications.
Faster and/or high dose escalation
Patients using fentanyl often have more opioid receptors in the brain “occupied” and adapted to stronger opioids, so a lower buprenorphine dose may not be enough to stop withdrawal and cravings.
If a patient still feels significant withdrawal and cravings after a “standard” starting dose, clinicians may increase the dose more quickly and reach a higher overall dose to control symptoms.
2025 Product Label Changes
For years, too many clinicians treated 16 mg or 24 mg as a hard ceiling.
In 2025, product labeling was officially updated so these numbers are not misread as maximum doses. FDA specifically warns that some patients need more than 24 mg/day.
High Dose Buprenorphine
A 2024 study in the journal Addiction found that increasing from 24 mg/day to 32 mg/day was associated with better outcomes (including improved retention and reduced opioid use).ⓘ
In practice, “high-dose” often means between 24-32 mg/day in an outpatient setting or even higher in rare inpatient cases.
If a patient is still in withdrawal or still craving opioids on a “standard” dose, the risk of dropping out of treatment goes up. With fentanyl in the mix, patients come in with higher tolerance and more severe withdrawal risk than when buprenorphine first became common.
Long-acting injections may have an advantage
A 2024 analysis reported that extended-release injectable buprenorphine may be more effective than sublingual buprenorphine/naloxone at reducing opioid use among participants with fentanyl use.ⓘ
New “Injectable-Only Overlapping” Start Protocol
A 2025 study in JAMA Network looked at a new protocol called “injectable-only overlapping” that uses a short series of long-acting injections up front, without requiring patients to stop fentanyl first or use buprenorphine tablets.ⓘ
The protocol is promising for those terrified of stopping fentanyl and reduces reliance on daily medication during the most fragile time. It does not require a patient to stop fentanyl use before starting, but insurance logistics may be a barrier, some withdrawals still occur, and further research is needed, especially on long-term abstinence.
In 2022, the FDA warned that buprenorphine medicines that dissolve in the mouth (under the tongue or against the cheek) have been linked to serious dental problems, including cavities, tooth decay, and tooth loss.ⓘ
Issues can happen even in people with no prior dental issues, but can be prevented by:
Buprenorphine remains a life-saving medication for opioid use disorder, and preventable dental side effects rarely outweigh the benefit of stopping full opioids.
While many states have not made changes impacting buprenorphine (including FL and CT), other states have improved or changed access either directly or indirectly. Examples below.
Opioid detox is the first step. Medications like buprenorphine help you get through the first days safely without getting pulled back to using.
In a medically supervised detox, the clinical team monitors symptoms, checks vitals, and uses medications to reduce withdrawal and cravings. It’s nothing like doing it on your own!
Get answers about your cost / coverage now.
Your information will be kept private
Let’s talk about what’s going on — no judgment. (We’ve been there before ourselves). No one will know you inquired and there is no commitment to call.
Need someone to talk to? We’re always here—day or night.
Ask questions, get guidance—no pressure, no obligation.
Your story stays with us. Confidential support, always.

Why Wait?
Get Answers Now
You’re here because you know you need help. Let’s talk through it together. There’s no commitment and it’s 100% confidential even to check your insurance.
100% Confidential